The EU vaccination campaign: a rough test for Europe’s strategic autonomy
So far, the vaccination campaign has been anything but the success story the European Union had hoped for. There are responsibilities for this in Brussels but also complex industrial and political realities that explain the current predicament.
The bloc is trailing Israel, the United Kingdom and the United States, which are leading on vaccinations on a global scale, having inoculated between eight and three times more than the EU. “This is the best advert for Brexit,” newspapers wrote in January on both shores of the Channel, as the immunization plan of the EU’s former member UK gathered pace.
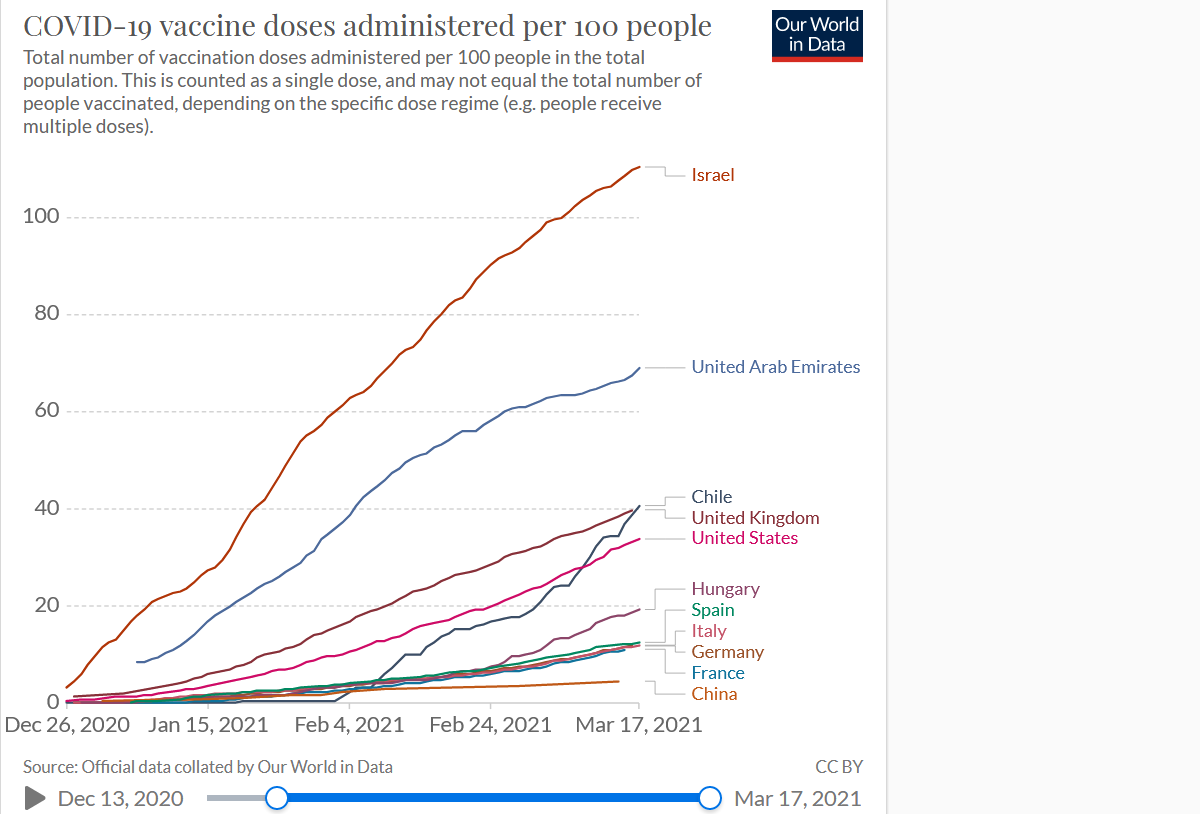
In the meantime, the vaccine situation has generated mounting frustration among national leaders and citizens given the shortfalls in dose deliveries, growing fears about possible side effects, and the geopolitical tensions surrounding the global vaccine supply chain.
Against this backdrop, the European Commission has become the scapegoat for all drawbacks, from the bottlenecks in the continent’s own production, to the more rigorous authorization procedures in comparison with those of other nations. This mix resulted in a slower rollout in the EU, further delayed by the de facto export ban enforced by key allies.
Confronted with competition from the Russian and Chinese shots, which have been increasingly appealing in certain countries, as well as the rising doubts about the safety of the vaccine manufactured by the British-Swedish company AstraZeneca, the EU is now struggling to take a clear stance to prevent member states from breaking the ranks.
Whether coordinating common positions in pairs or in small groups, EU countries have shown little consistency with the plan jointly developed with the Commission, massively diverging from the mainstream strategy and narrative agreed at the European level, and even blaming Brussels for the slow vaccination campaign and for shortcomings in the national deployment of doses.
Time is of the essence, however, to mantain the public trust in the vaccines, as well as the EU’s political credibility and ultimately its future. A more assertive leadership is essential to avoid fragmentation and to uphold a scientific evidence-based approach vis-à-vis restless member states, on the one hand, and to rescue global health cooperation, on the other.

The bloc’s strategy
On paper, the EU’s joint procurement scheme is sensible.
Last summer, the EU decided to negotiate vaccine purchases as a bloc, with the Commission acting on behalf of the member states, with several countries involved in the management and all 27 approving the various deals before the signature. This means that the EU has been able to buy vaccines at more convenient prices than many other Western countries. It has also diversified its vaccine portfolio, securing doses from six different producers.
To date, four of them have been authorized to provide vaccines for use following positive scientific recommendations by the European Medicines Agency (EMA), which granted conditional market authorizations to Pfizer/BioNTech (21 December), Moderna (6 January), Oxford/AstraZeneca (29 January), and the single-shot Johnson&Johnson (11 March). Two additional contracts have already been concluded with Sanofi/GSK and CureVac, while exploratory talks are ongoing with Novavax and Valneva.
Whereas the Commission has not publicly considered the inclusion of the Russian vaccine Sputnik V in its portfolio, EMA has started a rolling review of the vaccine, following the application by a German pharma manufacturer. Several key member states are now pushing for the EUs to buy and produce at home doses of Sputnik, whose positive reviews have been recently published on The Lancet. Some others, especially the Baltics and Poland, are opposing the move. It would represent an unexpected and undesirable pro-Moscow twist at a time when even light cooperation has been halted by the detention of political opponent Alexey Navalny, on top of the Crimea occupation and the meddling in the business of the countries of the Eastern Neighborhood.
The green light by the EU’s regulator is necessary but not sufficient for a shot to be included in the EU’s strategy, the Commission has recently warned, adding that it is ultimately up to the bloc’s executive and countries to enlarge the portfolio.
In the meantime, member states can, however, still partially depart from the EU’s common approach and grant national emergency authorizations to vaccines other than those approved by the EMA, as done by Hungary with Russia’s Sputnik V and China’s Sinopharm. After having stormed the final approval of Next Generation EU – the bloc’s recovery package -, threatening a veto for the linkage with the respect of the rule of law, Viktor Orbán has now thrown the gauntlet to Brussels once again, questioning the very existence of a common and effective European strategy.
The Hungarian leader is not alone in undermining the EU’s plan for domestic political purposes. Austrian Chancellor Sebastian Kurz traveled to Israel to strike a deal with his Danish counterpart and Prime Minister Benjamin Netanyahu on the joint development of second generation vaccines able to counter mutations of the virus, just a few days after the Commission had announced its own plan to adapt existing vaccines to the fast-spreading variants.
Afterwards, Chancellor Kurz began leading a group of small- and medium-sized member states that are bashing the Commission because of alleged “huge disparities” in the allocation of vaccines among the bloc’s countries. While contracts were negotiated and concluded by the EU, however, member states were left free to determine their own procurement choices, as not all shots have the same nominal price.
Since the beginning, the composition of the national vaccination portfolios and the quotas per each of the six authorized shots has been the sole responsibility of the national governments.
If industrial refrigerators are stockpiled with COVID-19 vaccines that are not administered, this is correctly blamed on the national plans, not the EU supply strategy.
Production bottlenecks and export disputes
What the EU might have fully realized only after the start of its massive vaccination campaign at the end of 2020 is the dire need to scale up production capacity on the continent to keep pace with the self-declared objective of inoculating 70% of the adult population by the end of the summer (22 September). Just a handful of industrial plants have been expressly indicated in the contracts as the sites where to produce the doses.
Some non-EU factories have been included in the agreements as authorized manufacturers and suppliers from outside the bloc: this is the case, for example, of two plants in the UK and one in the US, which all produce AstraZeneca shots.
However, both countries have some de facto export ban in place to make sure that American and British citizens are vaccinated first. This has contributed to the delay the delivery of doses to the EU, on the one hand, and further tightened the post-Brexit confrontational climate between London and Brussels amid accusations of vaccine nationalism, on the other.
At the beginning of March, in a move to show renewed trust in international cooperation, the EU released the numbers of the vaccine exports made since February under a new authorization export control scheme put in place as a measure to ensure that manufacturers would not breach any contractual engagement with the EU. So far, only one export request has not been granted – that of some 250,000 AstraZeneca shots from Italy to Australia -, while 41 million doses have been sent to 33 third countries, with the UK being the main beneficiary, according to recent data. On 17 March, European Commission President Ursula von der Leyen insisted on the need for reciprocity as “open roads run in both directions”, adding that Brussels might reconsider exports to countries with higher vaccination rates than the EU.
The vast majority of the exported doses, which made part of the public opinion question the openness of the EU while the bloc’s vaccination campaign continued to sputter on, come from the facilities of Pfizer/BioNTech, which is honoring its contract with the EU and has also increased its planned deliveries for the months to come.
It is worth noting that these figures do not include the deliveries to countries of both the Eastern and Southern Neighborhood, as well as those in the Balkans and to the COVAX scheme of the United Nations – the coalition to ensure that 92 middle- and lower-income countries have equal access to COVID-19 vaccines, of which the EU is the main donor. It would be short-sighted to question them now, for a Union that wants to be a “geopolitical” actor.
Leading from Europe?
The race for vaccines is a global matter. The EU has been collectively vocal about safeguarding international cooperation, which is part of Europe’s DNA. Not to forget is also that key ingredients for vaccines adopting the mRNA technology, such Pfizer/BioNTech and Moderna, need components that are imported from third countries and that would be hindered by rising vaccine protectionism.
Despite all the difficulties, the clash on vaccines gives Brussels the opportunity to make the case once again for the EU’s strategic autonomy, intended as the ability to act with others, while being prepared to act alone. It is up to the EU to take action: internally, by recreating trust among its countries on a coherent and centrally managed vaccine procurement; externally, by salvaging international cooperation, starting with transatlantic ties and humanitarian commitments.
In the end, it is about shaping the world we will inherit once the pandemic is over.
